Valve2022
Valve2022 valve connects two nodes. |
Documentation
Valve2022 valve connects two nodes.
Parameters
- adaptation_Aopen_fac: double
factor used
- A_open: float
Opening area
- A_leak: float
Leaking valve area
- l: float
Length of valve
- rho_b: float
Blood density
- papillary_muscles: bool
If true, papilary muscle implementation is activated
- soft_closure: bool
If true, a soft closure is activated
Signals
- q: float
Flow through the valve
- qDot: float
Time derivative of the flow q
Physiological background
In a normal adult, the heart has four chambers, i.e., the left (La) and right atrium (Ra) and the left (Lv) and right ventricle (Rv). The heart valves are the orifices which connect atrium to ventricle and ventricle to artery, or also known as atrio-ventricular and ventriculo-arterial valves, respectively. Besides the latter valves, the atrial myocardial walls contain other (permanently open) orifices connecting the systemic veins with the right atrium and the pulmonary veins with the left atrium.
Atrio-ventricular (AV) and ventriculo-arterial valves
There are two atrio-ventricular valves, i.e., the mitral valve separating the La from the Lv, and the tricuspid valve, separating the Ra from the Rv. The two ventriculo-arterial valves are the aortic valve, separating the Lv from the aorta, and the pulmonary valve, separating the Rv from the pulmonary artery.
Opening of the valves is driven by a transvalvular pressure drop. During diastole, the atrioventricular valves are open, allowing the ventricles to be filled with blood. When the pressure in the ventricles exceeds that in the atria, these valves will start to close. This closure will occur gradually due to inertia of the accelerated blood volume. Similarly, when ventricular pressure exceeds that in the distal arteries, the ventriculo-arterial valves open and blood leaves the ventricles, reducing the volumes of the ventricles. The atria are in open connection with the venous circulation, allowing continuous flow into the atria.
Atrio-ventricular valve prolapse and regurgitation during systole are prevented by papillary muscle contraction. The LV papillary muscles attach the two cusps of the mitral valve to the anterior and posterior LV free wall. The RV papillary muscles attach the three cusps of the tricuspid valve to the anterior and posterior RV free wall and the septum. Normally, the papillary muscles are activated simultaneously with the ventricular myocardium and maintain tension throughout the entire systolic phase, thus preventing backflow by prolapse of the AV valves, which are forced back into the atria by the high pressure in the ventricles.
Pathophysiology and abnormal physiology
There are pathophysiological situations caused by congenital heart defects that result in extra openings in the heart. The atrial septal defect (ASD) and the ventricular septal defect (VSD) are orifices connecting the left atrium with the right atrium and the left ventricle with the right ventricle, respectively. During fetal development, there are other shunts like the foramen ovale (FO) and the ductus arteriosus (DA). The foramen ovale is a orifice connecting the right atrium with the left atrium like the ASD, whereas the ductus arteriosus connects the pulmonary artery with the descending aorta.
Valvular stenosis is an abnormal (disease) condition, in which the tissues forming the valve leaflets have become stiffer and thereby reduce the normal valve opening area (narrowing). As a result, forward blood flow through the valve is restricted so that the proximal cardiac chamber needs to work harder to eject the same amount of blood. Valvular insufficiency is an abnormal (disease) condition, in which the valve leaflets do not close properly, so that blood can flow backward (leak) across the valve. The process of backward blood flow often called “regurgitation”, “leakage” or “regurgitant flow”.
In a heart with asynchronous activation of the ventricular walls (due to a conduction disorder or pacing), the activation of the papillary muscles may be delayed, so that valvular prolapse and so-called “functional regurgitation” can occur.
The Valve2022 Module
In CircAdapt, all aforementioned orifices, either the physiological ones (the valves and venoatrial orifices) or the pathophysiological ones (ASD, VSD and FO) are modeled using the valve module. Therefore, the valve module in CircAdapt is used in a broader sense. In other words, the valve module is used to model any narrow orifice in the circulation where passing blood flow causes a pressure drop related to inertia and Bernoulli effect. Thus, the valve module is used to model both physiologic and pathophysiologic orifices . The valve module can connect any pair of nodes, associated with any Chamber, TriSeg, ArtVen and/or Tube module. Examples of these combinations are chamber-to-tube (aortic and pulmonary VA valves), tube-to-chamber (veno-atrial inlets), chamber-to-chamber (mitral and tricuspid valves, ASD and VSD), and tube-to-tube (systemic-to-pulmonary artery shunt).
The state variable in this module is blood flow q through the valve. The module adds an ordinary differential equation dq/dt == f(q,Δp) to the system of equations solved in CircAdapt.
Physical background
The Navier-Stokes equation describes the relation between pressure p and flow velocity v in a flow field of an incompressible fluid:
where symbols ρ and η refer to blood density and viscosity, respectively. In a cylindrical tube, assuming laminar flow and dominance of flow velocity vz in the axial z-direction, Eq. (1) is simplified to:
Integration along a stream line, neglecting viscosity and the effect of gravity over the short distance within the valve, for the pressure difference Δp over a trajectory it follows:
For steady state flow, proximal to the valve orifice streamlines are converging (Fig. 3) in a practically laminar fashion, allowing application of Eq. (3). However, distal to the orifice, streamlines diverge, generally resulting in vortices and turbulence with loss of energy. In the valve module we assumed that no pressure is regained by deceleration of the blood distal to the orifice.
Model Description
The valve in the CircAdapt model is based on Eq.(3). For proper incorporation in the model velocity vz is not directly available. Instead we used the state variable flow q. The following assumptions and simplifications were applied:
Blood is an incompressible, Newtonian fluid
The effect of gravity is neglected
Distal to the valve orifice, kinetic energy Ekin is lost to the level of exit velocity
Inertia L is estimated using the equation Ekin=½Lq2
A representative value for velocity is obtained by flow q divided by effective area A
At the inlet side all pressure-flow energy is converted to kinetic energy
At the outlet side, pressure is not regained even though velocity is decreasing, implying loss of energy to be attributed to turbulence and viscous losses.
As a first approximation we assume there is plug flow at every location in the valve, i.e., there is no gradient in flow velocity perpendicular to the stream lines.
Given a steady state flow q, the pressure difference Δp over the valve, as sketched in Fig. (.), is determined by the Bernoulli equation, as shown in Eq. (.) without the time-dependent term:
The first term refers to the proximal side of the valve, where laminar flow without energy loss is assumed to hold. The second term considers regaining of pressure in a divergent flow field. We assumed there is no regain, implying fac=0. If some pressure is regained, i.e., fac>0, we may assume an effective valve cross-section Aeff in stead of Avalve. Note that Δp is negative, since in general it holds Aeff<Aprox. Now consider flow dynamics, total kinetic energy Ekin equals the volume integral of ½ρv2 over the whole valve and its sphere of influence. Assuming that velocity is the same within each section, and equal to flow q divided by area A, it follows:
Using Ekin = ½ L q2,
In this equation all length and area parameters are condensed to the ratio of an effective length leff, and an effective area Aeff as determined above for steady state flow. Flow velocity is known not to be uniform. E.g., flow entering an orifice is contracting towards the center of the orifice, making the effective orifice Aeff to be smaller than the physical orifice size. Furthermore. Inertia L is known to depend on the distribution of flow velocity within the valve region. By choosing an appropriate value for leff the effect of inertia can be approximated quite well, while the number of valve parameters remains limited. So, in the final version we get:
Note: For an orifice with negligible length, the effective length leff is quite well approximated by the diameter of the orifice.
The characteristics of a valve depend typically on the flow direction. If flow reverses, in Eq. 3-7) we should interchange proximal and distal. For regular valves, forward and backward flow behavior is very different. With forward flow Aeff is much larger than with backflow. In the implementation, Aeff is always kept positive to avoid zero-division. The value of leff is less critical for change of flow direction, and is therefore kept constant. The CircAdapt program requires the time derivative of flow dq/dt to be written explicitely as a function of the pressure drop pdrop, being the negative of Δp. Rewriting of Eq.(7) renders:
Valve opening and closure
Obviously a regular valve closes if flow q reverses. However, if the pressure drop pdrop acts in the reverse direction, the valve remains open, as long as flow is forward. In the aortic valve, such situation occurs in the late phase of systole. Then, aortic flow decelerates while aortic pressure exceeds left ventricular pressure. Valve closure occurs if flow changes sign. On the other hand, if the aortic valve is closed, flow velocity through the very small leaking orifice area is very high. If pressure reverses, according to the model, the leakage flow needs time to decelerate due to the inertia of the narrow flow channel. In the real situation, because of the very flexible valve leaflet material the aortic valve opens immediately at the moment the pressure drop becomes positive. So, the used criterion for valve opening and closure is as follows:
if q>0 OR pdrop>0 → open
if q<0 AND pdrop<0 → closed
With open valve, orifice area Aeff is set to Aopen and with the closed valve to Aleak.
Pappilary Muscle Function
The Valve2022 module has optional papillary muscle function. If ‘papillary_muscles==False’, the parameter ‘Aleak’ is used to describe leaking area during back flow. If ‘papillary_muscles==True’, effective Aleak is described by:
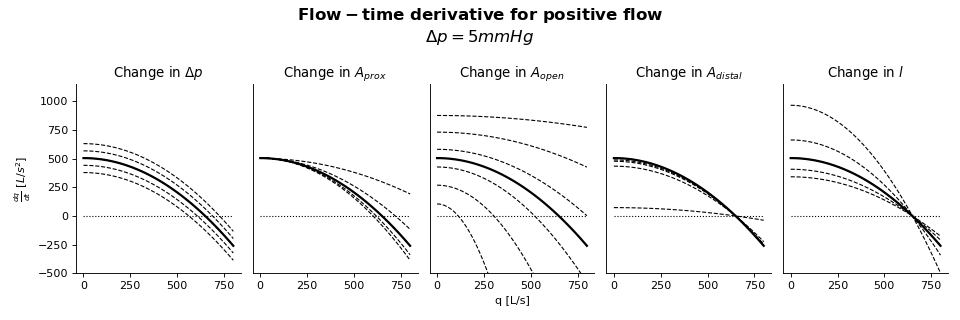
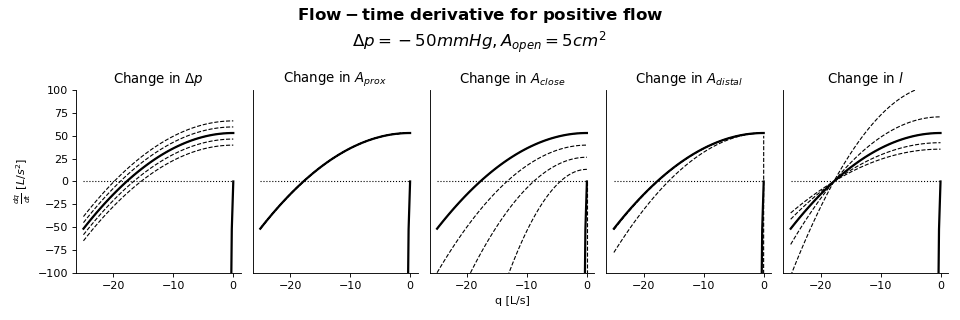
Sensitivity Analysis